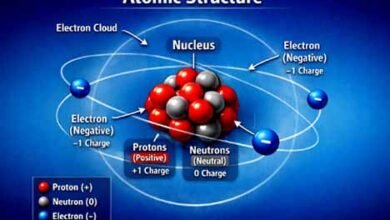
| One gm –mole of Heat S.T.P. = 22.4 lit Case 2:- molar volume at R.T.P = 24.45 lit One gm-mole oxygen at R.T.P = 22.45 lit One gm-atom of Heat R.T.P =22.45 lit Unit of volume M3 ,dm3, cm3 1m = 10dm = 100cm 1m3 =1000m3 ( lit ) = 106cm3(cc) 1m3 = 1000lit 1lit = 1000cc = 1000ml Density:- Mass per unit volume is called density Density = mass/volume = Unit = gm/cc, gm/lit, kg /m3 D = 1010kg /m3 =1010x1000gm/1000lit = 1010gm / it =1010gm/1000cc = 1.010gm/cc Density of H2o d = 1gm / cc Question:- calculate the total no of atoms in 11.2 lit f oxygen of oxygen at STP Solution:- One gm-mole oxygen = 32gm = 22.4lit = 3.011x1023mlecular 11.2 lit = 3.011×1023 1molecular = 2atom = 3.011×1023 x2 = 6.022×1023 Measurable properties of gas· ( 1) Mass Unit = gm, kg , mole (2) volume Volume of gas = volume of vessel M =VX d Unit of volume = m3,dm3(lit) cm3(cc) |